Osha infectious disease standard

The Occupational Safety and Health Administration (OSHA) is developing a proposed infectious disease standard aimed at protecting workers from biological hazards in various workplace settings. This long-anticipated regulation seeks to establish comprehensive requirements for exposure control, personal protective equipment, training, and emergency preparedness.
Targeting high-risk sectors such as healthcare, emergency services, and laboratories, the standard could significantly enhance worker safety during outbreaks of diseases like influenza, tuberculosis, and emerging pathogens such as SARS-CoV-2.
By creating uniform safety protocols, OSHA aims to reduce occupational infections and improve public health outcomes, marking a critical step toward safeguarding frontline workers in the face of ongoing and future infectious disease threats.
 America racial discrimination
America racial discriminationUnderstanding the OSHA Infectious Disease Standard and Workplace Safety
The Occupational Safety and Health Administration (OSHA) does not currently have a single, comprehensive standard specifically titled the Infectious Disease Standard. However, OSHA addresses the prevention and control of infectious diseases in the workplace through a combination of existing regulations, guidelines, and general duty obligations.
The agency’s approach focuses on minimizing employee exposure to biological hazards, including viruses, bacteria, and other pathogens, particularly in healthcare, laboratories, emergency response, and other high-risk sectors.
Key components of OSHA’s framework include the Bloodborne Pathogens Standard (29 CFR 1910.1030), the Respiratory Protection Standard (29 CFR 1910.134), and the General Duty Clause of the Occupational Safety and Health Act, which requires employers to provide a workplace free from recognized hazards.
During public health emergencies—such as the H1N1 pandemic or the COVID-19 outbreak—OSHA issues enforcement guidance, temporary emergency standards, and compliance directives to help employers protect workers from infectious agents. Effective infection control relies on hazard assessment, personal protective equipment (PPE), engineering controls (e.g., ventilation, isolation rooms), administrative controls (e.g., sick leave policies, training), and vaccination programs, where applicable.
 American airlines racial discrimination lawsuit
American airlines racial discrimination lawsuitKey Regulatory Frameworks Under OSHA for Infectious Disease Control
OSHA’s efforts to combat infectious disease in the workplace are grounded in several core regulations. The most directly applicable is the Bloodborne Pathogens Standard, which protects workers from exposure to blood and other potentially infectious materials (OPIM) through requirements like the development of an exposure control plan, use of engineering and work practice controls, provision of PPE, and offering hepatitis B vaccinations.
The Respiratory Protection Standard becomes crucial when airborne transmission is a concern, mandating the use of NIOSH-approved respirators and comprehensive respiratory protection programs, including fit testing and training. In addition, OSHA’s Personal Protective Equipment (PPE) Standard requires employers to assess workplace hazards and provide appropriate protective clothing and equipment.
The Hazard Communication Standard ensures that employees are informed about hazardous chemicals, including disinfectants used against pathogens. Lastly, the General Duty Clause empowers OSHA to cite employers for exposing workers to serious infectious disease risks even in the absence of a specific standard, provided that the hazard is recognized, foreseeable, and likely to cause harm.
Industry-Specific Applications and High-Risk Workplaces
Certain industries face significantly higher risks of exposure to infectious diseases, and OSHA's guidance is tailored accordingly. Healthcare settings—including hospitals, clinics, and long-term care facilities—are among the most regulated due to frequent contact with infected patients and contaminated materials.
 An example of racial discrimination
An example of racial discriminationEmergency medical services (EMS), correctional facilities, laboratories, mortuaries, and wastewater treatment plants also fall under heightened scrutiny. In these environments, exposure control plans must be site-specific and regularly updated.
For instance, a hospital may implement isolation protocols, needleless systems, and antiretroviral post-exposure prophylaxis (PEP), while a laboratory may emphasize biosafety levels (BSLs), autoclaving, and biosafety cabinets.
OSHA emphasizes the importance of worker training on infection transmission routes, proper decontamination procedures, and the correct use of PPE. In recent years, non-healthcare sectors such as meatpacking, retail, and public transit have also received attention during outbreaks, highlighting the need for cross-sector preparedness and flexible safety protocols.
Emergency Response and Temporary Standards During Outbreaks
During widespread infectious disease outbreaks—such as the 2009 H1N1 influenza pandemic or the 2020–2023 COVID-19 pandemic—OSHA has activated emergency measures to safeguard workers.
 Anti racial discrimination
Anti racial discriminationIn June 2021, OSHA issued an Emergency Temporary Standard (ETS) for COVID-19 specifically for healthcare settings, establishing detailed requirements for screening, physical distancing, ventilation, PPE, and outbreak response.
Although this ETS was later rescinded as the public health emergency ended, it demonstrated OSHA’s capacity to rapidly implement binding protections during crises. The agency also released voluntary guidance for non-healthcare sectors, encouraging employers to adopt infection prevention strategies like remote work, symptom screening, and enhanced cleaning.
Moreover, OSHA conducts targeted inspections in high-hazard workplaces during outbreaks and prioritizes complaints related to infectious disease exposure. The development of a permanent OSHA standard for infectious diseases has been discussed for years, with stakeholders urging a more proactive, unified approach to prevent future occupational health emergencies.
| OSHA Standard or Provision | Key Requirements | Applicable Industries |
|---|---|---|
| Bloodborne Pathogens (29 CFR 1910.1030) | Exposure control plan, PPE, training, hepatitis B vaccine, post-exposure evaluation | Healthcare, labs, emergency response, correctional facilities |
| Respiratory Protection (29 CFR 1910.134) | Respirator selection, fit testing, medical evaluations, training | Healthcare, construction, manufacturing, emergency services |
| Personal Protective Equipment (29 CFR 1910.132) | Hazard assessment, PPE provision and maintenance, employee training | All industries with exposure risks |
| General Duty Clause (OSH Act Section 5(a)(1)) | Employer obligation to address recognized serious hazards not covered by specific standards | All workplaces, especially during pandemics |
| Emergency Temporary Standard (ETS) for COVID-19 (Rescinded) | Screening, physical distancing, ventilation, PPE hierarchy, outbreak procedures | Healthcare and high-risk settings during the pandemic |
OSHA Infectious Disease Standard: A Comprehensive Compliance Guide
What does OSHA 1910.1450 cover in relation to infectious disease safety standards?
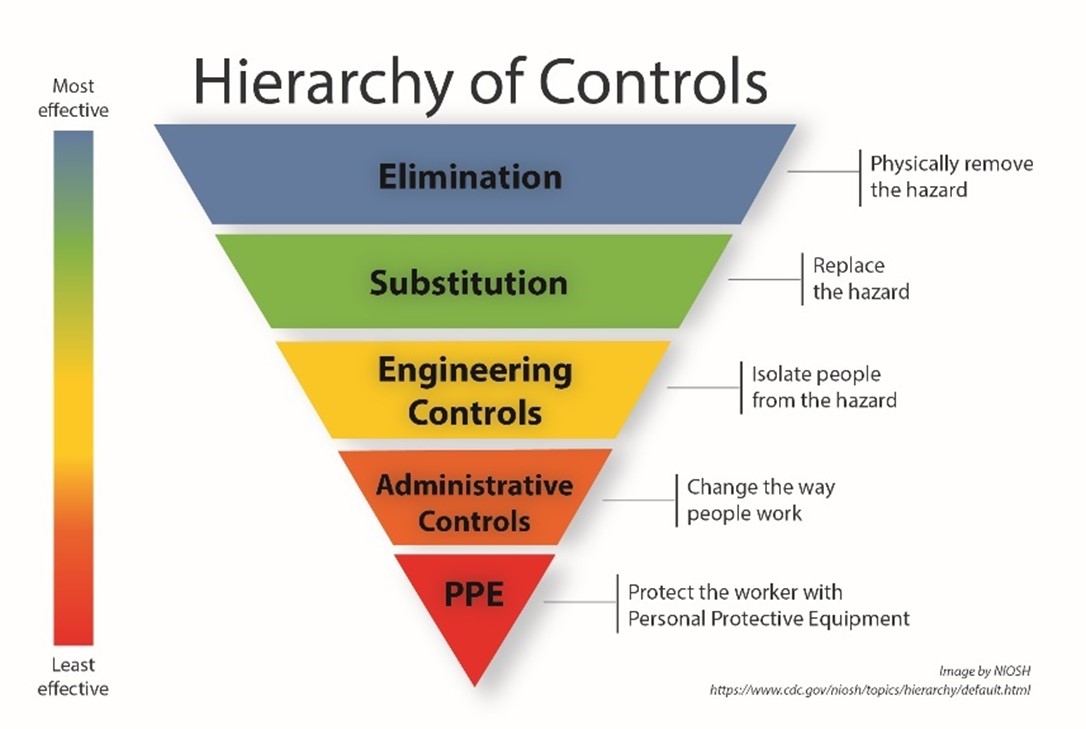
OSHA 1910.1450, titled Occupational Exposure to Hazardous Chemicals in Laboratories, is commonly known as the Laboratory Standard.
While it primarily addresses chemical hazards in laboratory settings, its principles can indirectly support infectious disease safety by promoting a culture of safety, proper use of personal protective equipment (PPE), and rigorous protocols for handling hazardous substances. However, it is important to clarify that OSHA 1910.1450 does not specifically regulate infectious agents or biological hazards such as viruses or bacteria.
Those are generally covered under other standards, including the General Duty Clause, guidelines from the CDC, and biosafety practices outlined in the Biosafety in Microbiological and Biomedical Laboratories (BMBL). Nevertheless, many laboratory safety practices promoted under 1910.1450—such as engineering controls, training, and exposure monitoring—can complement infection control measures when working with infectious materials in research laboratories.
Scope and Application of OSHA 1910.1450 in Laboratory Settings
- OSHA 1910.1450 applies to all employers engaged in laboratory use of hazardous chemicals, defined as chemicals that pose a physical or health hazard, including carcinogens, reproductive toxins, and substances with high acute toxicity.
- The standard requires the implementation of a Chemical Hygiene Plan (CHP), which outlines procedures, equipment, PPE, and work practices to protect workers from chemical exposure. While not designed for biological agents, these structured safety protocols can be adapted to include measures relevant to infectious disease prevention.
- Laboratories covered under this standard must ensure that employees receive specific training on hazard recognition, safe handling practices, and emergency procedures, elements that parallel the training needed when working with infectious agents.
Overlap Between Chemical and Biological Safety Practices
- Engineering controls such as fume hoods, biological safety cabinets (BSCs), and ventilation systems are emphasized in 1910.1450 for chemical containment and are also essential for preventing airborne transmission of infectious agents.
- The use of PPE—like gloves, lab coats, eye protection, and respirators—is mandated under the standard when handling hazardous chemicals and is equally critical for minimizing exposure to infectious materials.
- Requirements for proper labeling, storage, and disposal of hazardous chemicals support overall laboratory discipline, which helps maintain a clean and controlled environment conducive to infection control.
Limitations of OSHA 1910.1450 Regarding Infectious Agents
- OSHA 1910.1450 does not specifically address biosafety levels, handling of viable pathogens, or decontamination procedures for infectious waste, which are instead governed by CDC/NIH guidelines and OSHA’s Bloodborne Pathogens Standard (29 CFR 1910.1030).
- The standard does not require medical surveillance or vaccinations for exposure to infectious diseases, unlike the Bloodborne Pathogens Standard, which mandates hepatitis B vaccination and post-exposure evaluation.
- While it encourages exposure assessment and recordkeeping for chemical hazards, it lacks provisions tailored to monitoring biological exposure incidents, such as needlestick injuries or aerosol exposures to infectious materials.

Lack of a Written Exposure Control Plan
- One of the most frequent OSHA violations related to infectious disease standards is the failure to maintain a written Exposure Control Plan (ECP) as required by the Bloodborne Pathogens Standard (29 CFR 1910.1030). This document outlines procedures to protect workers from exposure to blood and other potentially infectious materials (OPIM).
- Employers are required to update the ECP annually and whenever changes in tasks, procedures, or employee positions affect or create new occupational exposure. Failure to revise the plan, even if initially compliant, results in citation.
- The ECP must include details such as exposure determination, procedures for evaluation of incidents, schedule for training, and methods of compliance like engineering controls and PPE. OSHA often finds plans either missing key elements or not being implemented effectively in practice.
Inadequate Training and Employee Information
- OSHA mandates that employees with potential exposure to infectious materials receive comprehensive training upon initial assignment and annually thereafter. A common violation occurs when employers fail to provide this training or do not keep accurate records of completed sessions.
- The training must cover topics such as modes of transmission of bloodborne pathogens, appropriate use of PPE, actions to take following an exposure incident, and information about the HBV vaccine. Incomplete or generalized training that doesn’t address site-specific risks is considered non-compliant.
- Additionally, workers must be informed about the location and availability of the Exposure Control Plan, as well as their right to medical evaluation after exposure. Lack of accessible information or failure to communicate these rights is frequently cited during inspections.
Failure to Use Universal Precautions and Provide Proper PPE
- A core component of OSHA’s infectious disease standards is the requirement to follow Universal Precautions, treating all human blood and OPIM as if they are infectious. Many violations arise when employees are not consistently applying these precautions, particularly in non-traditional healthcare environments.
- Employers must provide appropriate personal protective equipment (PPE), such as gloves, gowns, face shields, and masks, at no cost to employees. OSHA commonly cites workplaces for not supplying the correct type of PPE, allowing reusable PPE without proper cleaning, or permitting damaged or outdated equipment to be used.
- Moreover, failure to ensure proper use, removal, and disposal of PPE—along with inadequate hand hygiene practices—contributes to non-compliance. These lapses increase the risk of transmission and are significant factors in OSHA enforcement actions related to infectious disease control.
What OSHA regulations address infectious disease exposure in the workplace?
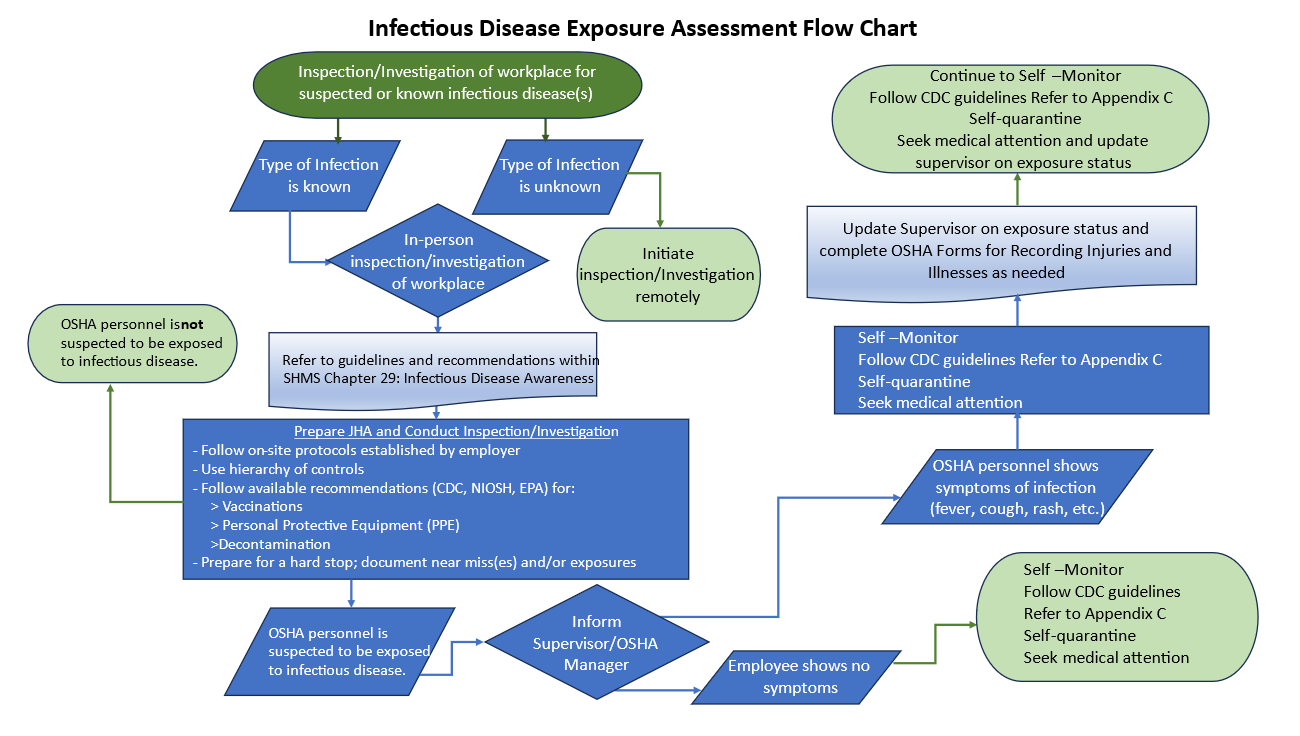
Bloodborne Pathogens Standard (29 CFR 1910.1030)
- The Bloodborne Pathogens Standard is one of OSHA’s primary regulations addressing infectious disease exposure, specifically targeting diseases transmitted through blood and other potentially infectious materials (OPIM), such as HIV, hepatitis B, and hepatitis C.
- This standard requires employers to develop and implement an Exposure Control Plan that outlines protective measures, including engineering controls (e.g., sharps disposal containers), work practices (e.g., no recapping of needles), and the use of personal protective equipment (PPE) like gloves and gowns.
- Employers must also provide annual training on bloodborne pathogens, offer the hepatitis B vaccine to at-risk employees at no cost, and ensure prompt post-exposure evaluation and follow-up in the event of an exposure incident.
Personal Protective Equipment (PPE) Standards (29 CFR 1910, Subpart I)
- OSHA’s PPE standards mandate that employers assess the workplace to determine where infectious disease hazards exist and provide appropriate protective gear, such as face shields, respirators, gloves, and protective clothing.
- These regulations require employers to train employees on the proper use, maintenance, and disposal of PPE, ensuring that it effectively reduces the risk of exposure to infectious agents.
- Employers are also responsible for cleaning, repairing, and replacing PPE as needed at no cost to the employee, which is crucial in healthcare, laboratories, and other high-risk environments.
General Duty Clause (Section 5(a)(1) of the OSH Act)
- When no specific OSHA standard applies to a particular infectious disease, such as during emerging outbreaks like pandemic influenza or novel coronavirus, the General Duty Clause requires employers to provide a workplace free from recognized hazards that could cause death or serious physical harm.
- This clause has been used to hold employers accountable for failing to implement infection control measures, including proper ventilation, social distancing, symptom screening, and providing face coverings or respirators during public health emergencies.
- OSHA may issue citations under the General Duty Clause if an employer knew or should have known about a significant risk of infectious disease exposure and did not take feasible steps to mitigate it.
Frequently Asked Questions
What is the OSHA Infectious Disease Standard?
The OSHA Infectious Disease Standard is a proposed regulation aimed at protecting workers from exposure to infectious diseases in the workplace. It would require employers to implement infection prevention and control measures, including exposure control plans, personal protective equipment, training, and vaccination programs. The standard seeks to reduce occupational risks associated with pathogens like viruses and bacteria, especially in healthcare and high-risk settings.
Which industries are most affected by the OSHA Infectious Disease Standard?
The OSHA Infectious Disease Standard primarily affects healthcare, emergency services, correctional facilities, and other high-risk workplaces where employees face frequent exposure to infectious agents. This includes hospitals, clinics, long-term care facilities, and laboratories. Workers in public transportation, waste management, and home healthcare may also be covered. The goal is to protect employees who are more likely to encounter bloodborne or airborne pathogens during their duties.
What are the key requirements under the OSHA Infectious Disease Standard?
Key requirements include developing a written exposure control plan, conducting hazard assessments, providing appropriate personal protective equipment (PPE), offering vaccinations (like flu and hepatitis B), training employees on infection risks, and implementing engineering and administrative controls. Employers must also establish procedures for exposure incidents, including medical evaluation and follow-up. These measures aim to minimize worker exposure and ensure a safer workplace during disease outbreaks.
Is the OSHA Infectious Disease Standard currently enforceable?
As of now, the OSHA Infectious Disease Standard is not fully adopted as a permanent enforceable regulation. OSHA has issued emergency temporary standards in specific situations, like the COVID-19 pandemic for healthcare, but a comprehensive, permanent standard is still under development. OSHA continues to evaluate risks and gather input to create a broad rule that would standardize protections against infectious diseases across various industries.

Leave a Reply